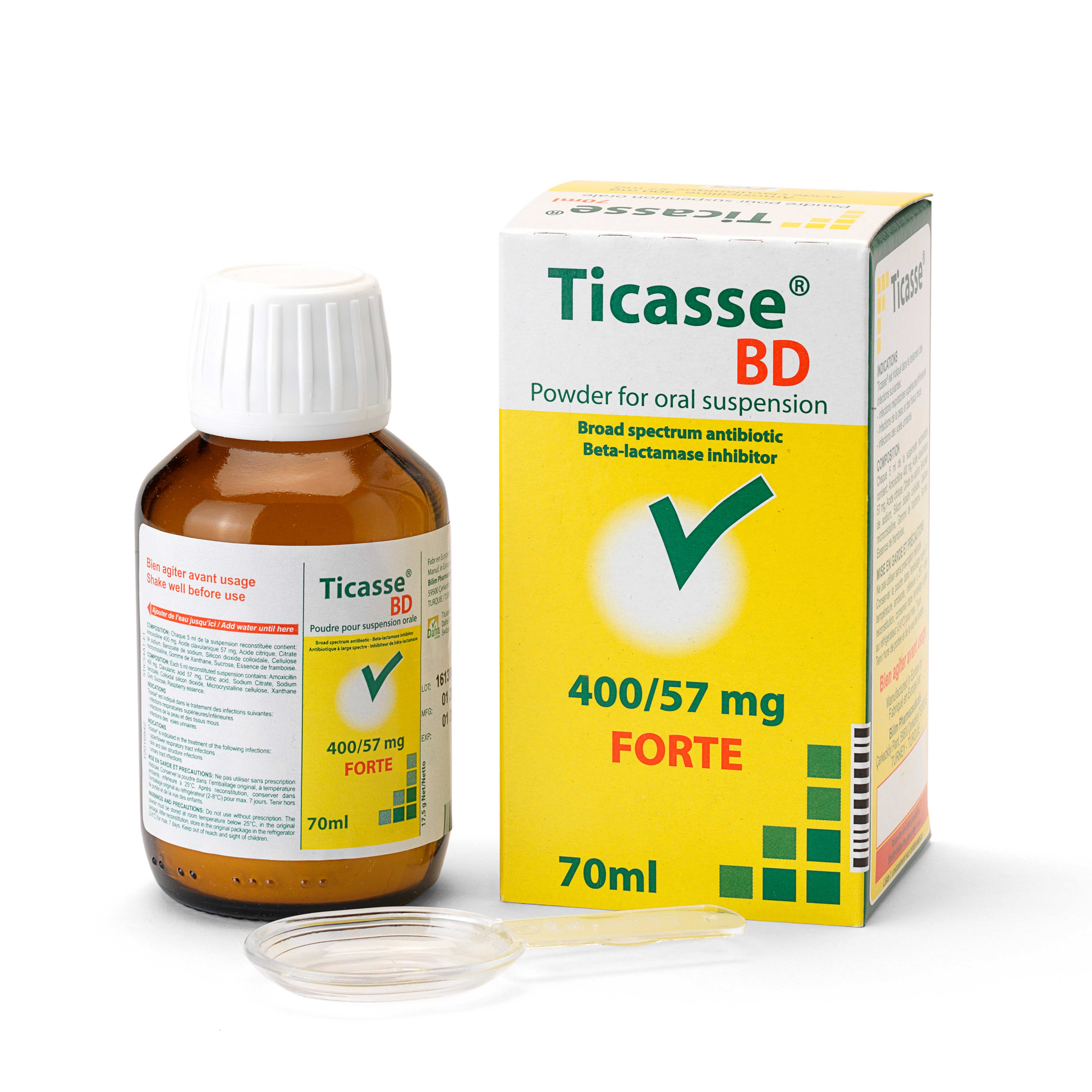
Active substance: amoxicillin – clavulanic acid
Form: powder for oral suspension
Dosage
Ticasse / Dafraclav 228: 200 mg amoxicillin and 28.5 mg clavulanic acid per 5 ml suspension
Ticasse / Dafraclav 457: 400 mg amoxicillin and 57 mg clavulanic acid per 5 ml suspension
Packaging:
– Bottle with powder for the preparation of 70 ml of oral suspension
Target:
Ticasse / Dafraclav 228: children up to 2 years (12kg)
Ticasse / Dafraclav 457: children from 2 to 6 years (12 to 30 kg)
Ticasse / Dafraclav suspension is a bactericidal antibiotic
– Amoxicillin rapidly kills susceptible bacteria
– Clavulanic acid prevents the inactivation of amoxicillin by enzymes produced by bacteria.
Ticasse/Dafraclav is indicated for the treatment of the following infections
Suspensions 200/28 and 400/57 per 5 ml: The usual dose is 25 mg to 45 mg of amoxicillin body weight per day.
The higher dose is up to 70 mg amoxicillin body weight per day.
The daily dose should be divided into two equal portions, administered every 12 hours.
Use the measuring spoon provided to give the appropriate dose.
Prescription drugs. Please contact your doctor.

| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |